Polar and nonpolar covalent bonds: characteristics & differences Polar covalent bond: definition and examples Covalent bonds nonpolar definition chemical description of a polar covalent bond
What is a Polar Covalent Bond? | ChemTalk
Covalent bonds formed chemistrylearner Nonpolar covalent bonds definition Polar and nonpolar molecules
Covalent bonds
Polar covalent bond: definition and examplesPolar covalent bonds Bond polarity electronegativity molecular shape covalent ionic bonding chemistry types atoms different figure between two polar nonpolar electron distribution electrons9.3: molecular shape and molecular polarity.
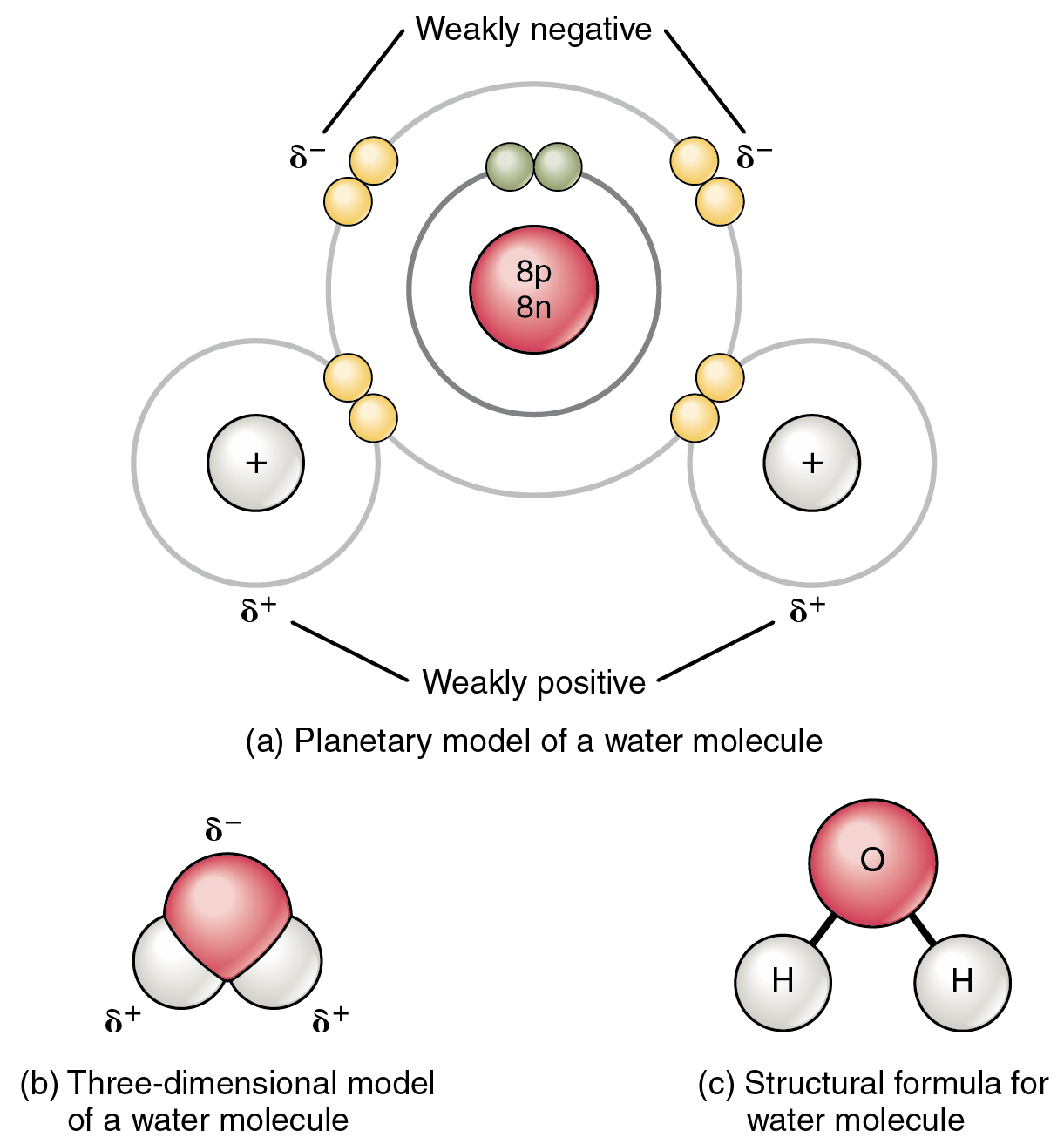
Double covalent bondBond polarity molecular electronegativity covalent ionic chemistry atoms types shape different between figure two polar nonpolar electron bonding electrons chemical Covalent bonds nonpolar bond polar molecule water molecules hydrogen shape biology three carbon figure atoms between molecular oxygen type dioxidePolar bond covalent chemistry examples chemical bonds definition molecule science bonding type types example molecules non nonpolar electronegativity between kids.

Covalent nonpolar polar bonds bond electrons
Covalent bondWhat is nonpolar covalent bond Definition and examples of a polar bondCovalent bonding (biology) — definition & role.
Polar covalent bonds acids and basesAttractive forces and bonds Polar covalent bonds chemistry acids basesCovalent bonds.

Covalent definition chemistry
Covalent bonding molecule electron oxygen atomWhat is a polar covalent bond? Covalent bond: definition, types, and examplesNonpolar covalent bond: definition and examples.
Polar covalent bond: definition and examples in chemistryBonds covalent attractive forces intramolecular atoms Polar nonpolar molecules bonding examples covalent molecule bonds sciencenotes induced chemical ionic organic intermolecular biology distributed evenly dipole9.3: shape and polarity.

What is a covalent bond in chemistry? aa3
Polar bond covalent bonds nonpolar chemistry ionic polarity electron properties electronegativity bonding electrostatic general vs between shape distribution lewis molecular .
.